You are here
Selenium Drives a Transcriptional Adaptive Program to Block Ferroptosis and Treat Stroke
PUBLICATION:
Journal ArticleYear Published:
2019Publisher:
Cell. 2019 May 16;177(5):1262-1279.e25. doi: 10.1016/j.cell.2019.03.032. Epub 2019 May 2.Identifiers:
PMID: 31056284 | DOI: 10.1016/j.cell.2019.03.032Abstract
Highlights
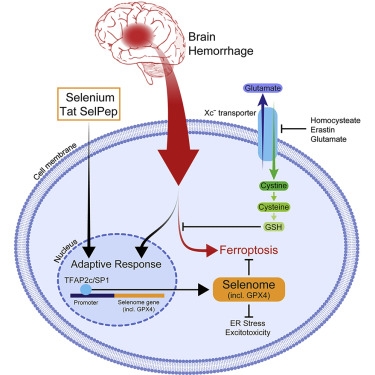
- Ferroptotic stimuli drive inadequate transcription of protective selenoproteins
- Selenium (Se) enhances adaptive transcription to protect cells from ferroptosis
- Se induces a protective selenome via DNA binding of TFAP2c and Sp1
- A selenocysteine-containing peptide, Tat SelPep, improves outcomes after stroke
Summary
Ferroptosis, a non-apoptotic form of programmed cell death, is triggered by oxidative stress in cancer, heat stress in plants, and hemorrhagic stroke. A homeostatic transcriptional response to ferroptotic stimuli is unknown. We show that neurons respond to ferroptotic stimuli by induction of selenoproteins, including antioxidant glutathione peroxidase 4 (GPX4). Pharmacological selenium (Se) augments GPX4 and other genes in this transcriptional program, the selenome, via coordinated activation of the transcription factors TFAP2c and Sp1 to protect neurons. Remarkably, a single dose of Se delivered into the brain drives antioxidant GPX4 expression, protects neurons, and improves behavior in a hemorrhagic stroke model. Altogether, we show that pharmacological Se supplementation effectively inhibits GPX4-dependent ferroptotic death as well as cell death induced by excitotoxicity or ER stress, which are GPX4 independent. Systemic administration of a brain-penetrant selenopeptide activates homeostatic transcription to inhibit cell death and improves function when delivered after hemorrhagic or ischemic stroke.