You are here
Translating Neurotrophin Biology to Accelerate Innovation in Psychiatry
Speakers
Abstract
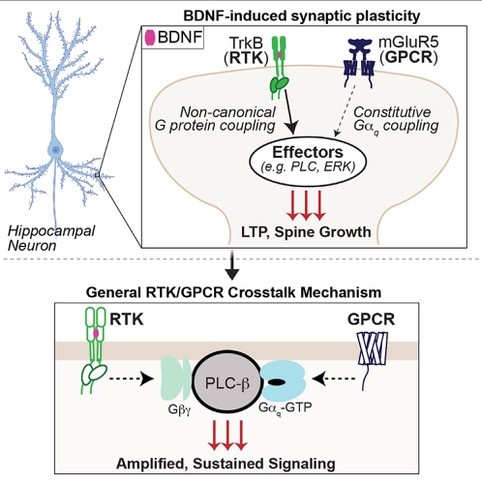
Synaptic plasticity is based on a core group of synaptic receptors that rapidly sense neurotransmitters such as glutamate and GABA. Neurotrophins, such as brain-derived neurotrophic factor (BDNF), are typically thought to signal on slower time scales, but have also been shown to regulate the induction and expression of synaptic plasticity. Despite both neurotransmitter and neurotrophin signaling underlying most forms of synaptic plasticity and serving as major mediators of the pathophysiology and treatment of neurogenerative and neuropsychiatric disorders, little is known about how these different receptor classes work in concert through direct and indirect forms of crosstalk. Recently, we found that the receptor tyrosine kinase (RTK) TrkB and the G-protein-coupled receptor (GPCR) metabotropic glutamate receptor 5 (mGluR5) together mediate hippocampal synaptic plasticity in response to brain-derived neurotrophic factor (BDNF). Activated TrkB enhances constitutive mGluR5 activity to initiate a mode switch that drives BDNF-dependent sustained, oscillatory Ca2+ signaling and enhanced MAP kinase activation. Collectively, this new form of TrkB-mGluR5 signaling may contribute to the diverse higher order nervous system functions related to neural plasticity that have been attributed to the BDNF-TrkB neurotrophin system.