You are here
Molecular Control of Neuronal Wiring: Plexin-A4 Signaling Specificity
Speakers
Abstract:
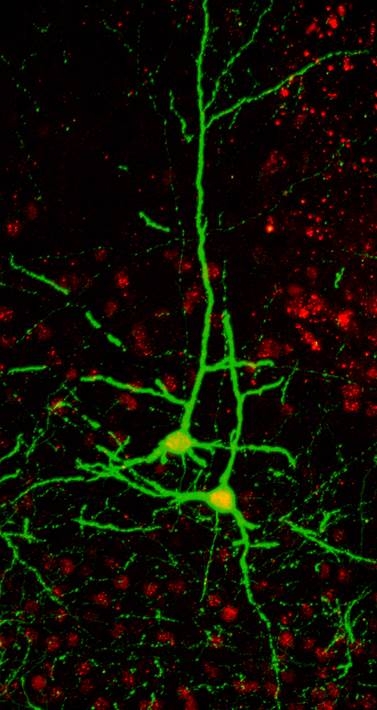
The primary focus of our research aims to investigate how proper formation of neuronal morphology influences the establishment of neuronal connections and functions in the mouse nervous system. During development the proper wiring of the nervous system is largely regulated by extracellular cues and their ability to induce diverse cellular morphogenesis through activation of receptors on the surface of developing neurons. The determinants for this diversity at the receptor level and its signal transduction are largely elusive. Semaphorin (Sema)3A signaling through the Neuropilin-1/Plexin (Plxn)A4 receptor complex had been demonstrated to promote basal dendrite arborization of cortical pyramidal neurons, and to repel CNS and PNS axons. How this same ligand-receptor pair achieved these distinct cellular processes in different populations of neurons is unknown. Using overexpression in vitro system we previously suggested that diverse responses elicited by Sema3A signaling could be found at the receptor level, within the cytoplasmic domains of PlxnA4. Here we generated a new PlxnA4 mouse line where the conserved triplet basic amino acids KRK, in the cytoplasmic domain of the receptor, were substituted with AAA. We found that this KRK-motif is required for Sema3A induced dendritic elaboration of cortical neurons both in vivo and in vitro. In contrast this motif is dispensable for growth cone collapse and axon repulsion of DRG sensory neurons in vitro and for inhibitory axonal guidance events of multiple neuronal cell types in vivo. Moreover, we show that the RhoGEF FARP2, which binds the KRK motif of PlexinA4, show identical specificity. Lastly, we also demonstrated that Rac1 activity is elevated in cortical neurons following Sema3A treatment and that Rac1 activation, by FARP2 upon Sema3A signaling, is required for dendrite elaboration but not for DRG sensory axon collapse. Our study uncovers a new pathway underlying Sema3A-Nrp1/PlxnA4 signaling that is specifically required to promote dendrite morphogenesis in vitro and in vivo. Broadly, our results show that the divergent signaling of multifunctional receptors can be pin-pointed to only three amino acids allowing for exploration of distinct cellular functions.